|
Cytological Laboratory | Niki Margari
Ampelokipi Attica
• Endometrial cancer is the most common malignancy of the female genital system. 75% of endometrial cancers are found in postmenopausal women, with a median age at diagnosis of 61 years.
• According to previously published data, the incidence of endometrial cancer ranges from 0.6 to 6/1000, so endometrial cancer can potentially be a serious public health problem, especially for postmenopausal women.
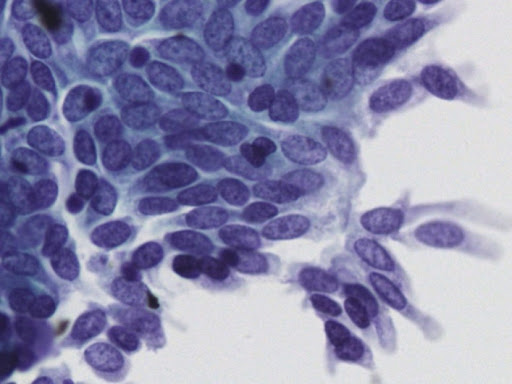
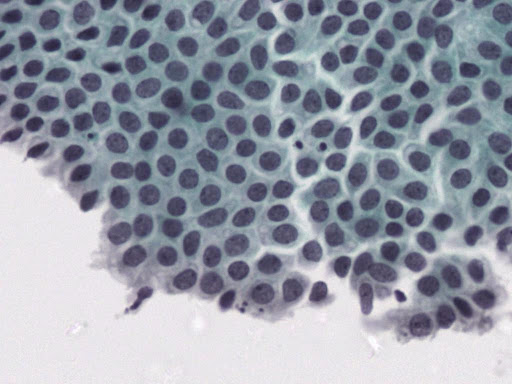
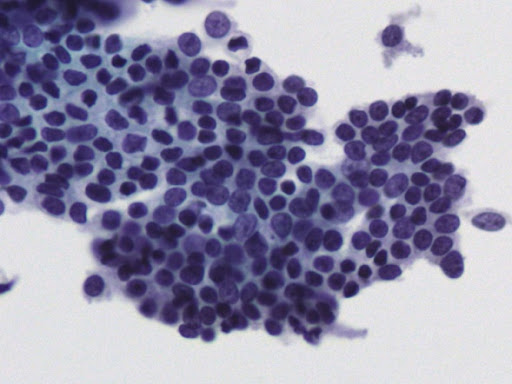
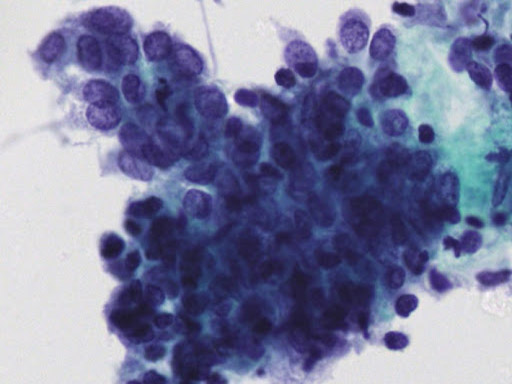
• Examination of material taken directly from the endometrial cavity is the most reliable diagnostic method for diagnosing endometrial cancer at the cytological level.
Today, endometrial cytology is recognized as an accepted diagnostic method in the United States, and since 1987 in Japan, following the Health Insurance Act for the Elderly, endometrial cytology has become a routine method of initial screening. of endometrial lesions.
• Previously published studies indicate that liquid phase cytology when combined with ultrasound examination has very good results.
SUITABILITY OF THE SAMPLE
The following parameters are important for evaluating the adequacy of the sampling process:
1. Necessary clinical information:
♦ history of menstruation,
♦ the use of birth control pills or other hormones or medications,
♦ the use of an endometrial coil,
♦ previous pathological cytological examination or biopsy,
♦ history of endometrial lesions and treatment followed
2. Immediate fixation of the sample after collection
3. Adequate presence of well-preserved cells in the sample
Bibliography:
1. The Yokohama system for reporting endometrial Cytology- Definitions, Criteria, and explanatory notes.
Book: Chapter 14, Springer 2022, ISBN 978-16-5010-9
2. The Yokohama system for reporting directly sampled endometrial cytology: The quest to develop a standardized terminology.
Fulciniti F, Yanoh K, Karakitsos P, Watanabe J, Di Lorito A, Margari N, Maeda Y, Kihara M, Norimatsu Y, Kobayashi TK, Hirai Y. Diagn Cytopathol. 2018 May; 46 (5): 400-412. doi: 10.1002 / dc.23916. Epub 2018 Feb 26. PMID: 29479846.
3. Internal quality control in an academic cytopathology laboratory for the introduction of a new reporting system for endometrial cytology.
Margari N, Pouliakis A, Aninos D, Meristoudis C, Stamataki M, Panayiotides I, et al.
Diagnosis Cytopathol. (2017) 45: 883–8. doi: 10.1002 / dc.23787
4. A reporting system for endometrial cytology: Cytomorphologic criteria-Implied risk of malignancy.
Margari N, Pouliakis A, Anoinos D, Terzakis E, Koureas N, Chrelias C, Marios Makris G, Pappas A, Bilirakis E, Goudeli C, Damaskou V, Papantoniou N, Panayiotides I, Karakitsos P. Diagn Cytopathol. 2016 Nov; 44 (11): 888-901. doi: 10.1002 / dc.23605.
Epub 2016 Sep 22. PMID: 27653446.
5. Office Endometrial Cytological Sampling: Examining Predictors of Strenuousness.
Makris GM, Siristatidis C, Margari N, Chrelias C, Papanota AM, Sergentanis TN, Karakitsos P, Papantoniou N. In Vivo. 2016 May-Jun; 30 (3): 309-14. PMID: 27107090.
6. A Pyrosequencing Assay for the Quantitative Methylation Analysis of GALR1 in Endometrial Samples: Preliminary Results.
Kottaridi C, Koureas N, Margari N, Terzakis E, Bilirakis E, Pappas A, Chrelias C, Spathis A, Aga E, Pouliakis A, Panayiotides I, Karakitsos P. Biomed Res Int.
2015; 2015: 756359. doi: 10.1155 / 2015/756359. Epub 2015 Oct 4. PMID: 26504828;
PMCID: PMC4609388.
7. Classification of endometrial lesions by nuclear morphometry features extracted from liquid-based cytology samples: a system based on logistic regression model.
Zygouris D, Pouliakis A, Margari N, Chrelias C, Terzakis E, Koureas N, Panayiotides I, Karakitsos P. Anal Quant Cytopathol Histpathol.
2014 Aug; 36 (4): 189-98. PMID: 25291856.
8. Using classification and regression trees, liquid-based cytology and nuclear morphometry for the discrimination of endometrial lesions.
Pouliakis A, Margari C, Margari N, Chrelias C, Zygouris D, Meristoudis C, Panayiotides I, Karakitsos P. Diagn Cytopathol.
2014 Jul; 42 (7): 582-91. doi: 10.1002 / dc.23077.
Epub 2013 Nov 22. PMID: 24273089.
|